Background: Primary central nervous system lymphoma (PCNSL) constitutes approximately 5% of extranodal lymphomas. More than 90% of PCNSL are diagnosed as diffuse large B-cell lymphomas (DLBCL), mainly classified as the activated B cell (ABC) subtype. Recent genetic analyses have identified key molecular drivers of PCNSL, including MYD88, CARD11 and CD79B. However, molecular analyses using bulk samples have provided limited insights into the heterogeneity in lymphoma cells of PCNSL. To improve our understanding in the biology of PCNSL, we conducted a multimodal single-cell analyses and integrated single-cell and spatial analyses to characterize the molecular properties of PCNSL tumors.
Methods:Our study included seven PCNSL patients and two patients with reactive lymph nodes (RLN). We performed Cellular Indexing of Transcriptomes and Epitopes by Sequencing (CITE-seq) combined with B-cell and T-cell receptor (BCR and TCR) analysis on samples from 3 PCNSL and 2 RLN patients, while the remaining PCNSL patients' samples were subjected to single-cell transcriptomic sequencing. Additionally, six formalin-fixed and paraffin-embedded (FFPE) tissues from the patients analyzed in the single-cell sequencing (except for one patient) were performed with GeoMx digital spatial profiling (DSP).
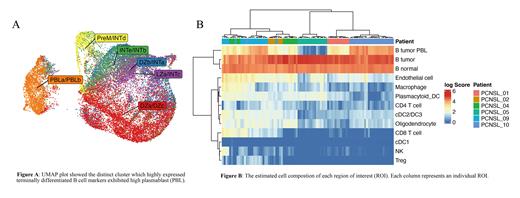
Result: To ensure data quality, we filtered out low-quality cells and doublets, resulting in a dataset of 41,556 single cells. B cells represented the most abundant cluster, followed by T cells and monocytes. Copy number alterations (CNAs) data derived from InferCNV analysis showed distinct CNAs patterns in lymphoma cells of each patient. UMAP plot projection analysis revealed the presence of a distinct subtype of malignant cells within the B-cell population, which was characterized as high expression of plasmablast (PBL)-associated surface proteins, including SLAMF7, CD39, and CD38. Enrichment analysis based on germinal center (GC) gene expression signatures (Holmes AB et al. J Exp Med. 2020) revealed a broader spectrum of B-cell differentiation states in malignant B cells. Single-cell BCR sequencing further demonstrated that different GC signature state cells harbored the same BCR repertoire within each patient, suggesting a common origin of malignant cells. Non-negative matrix factorization (NMF) analysis confirmed the enrichment of PBL signature scores in the distinct malignant cell population observed in the UMAP plot, consistent with DPE analysis (Figure A). Interestingly, the proportion of PBL signature cells varied among patients. Signaling interaction analysis highlighted CD70-CD27 and SPP1-CD44 pathways as enriched in tumor cells. PBL signature cells received TNFSF13B and TNFSF13 signaling through TNFRSF17 (BCMA), whereas other tumor cells expressed TNFRSF13C (BAFF-R) and received TNFSF13B signaling alone.
We further analyzed two public bulk RNA-seq data (Bruno A et al. Neuro Oncol 2018, n = 6; Radke J et al. Nat Commun 2022, n=38) in PCNSL to decompose tumor components. Tumor PBL components were identified in most PCNSL cases, although several cases exhibited a low presence of tumor PBL population. Of note, analysis including these public datasets with our single-cell cohort showed that PCNSL samples with high tumor PBL proportion had unfavorable prognosis .
Additionally, we performed DSP to integrate the single-cell and spatial multi-omics analyses. We deconvoluted the cell composition by using transcriptomic data derived from each region of interest (ROI) and found that the cell proportions in ROI were consistent with the corresponding single-cell results. However, there were differential profiles of cell composition according to patients rather than ROIs, suggesting that PCNSL tumors may display spatial homogeneity (Figure B).
Conclusion: Through the application of single-cell and spatial transcriptomic analysis, we identified a distinct subtype expressing the PBL signature gene. These findings have important therapeutic implications and provide new insights into the mechanisms of relapse in PCNSL.
Disclosures
Kobayashi:Chugai, Sanofi, Asahi Kasei Pharma, Janssen and Meiji Seika Pharma: Honoraria. Asada:Asahi KASEI Co., Ltd.: Speakers Bureau; Astellas: Speakers Bureau; Kyowa Kirin: Speakers Bureau; Meiji Seika Phama Co. Ltd.: Speakers Bureau; Nippon Shinyaku Co., Ltd: Speakers Bureau; Novartis: Research Funding, Speakers Bureau; Abbvie: Speakers Bureau. Fujii:Novartis: Honoraria; Janssen Pharmaceutical: Honoraria. Sato:Chugai Pharmaceutical Co., Ltd.: Honoraria; Kyowa Kirin: Honoraria; Takeda Pharmaceutical: Honoraria; Daiichi Sankyo: Current equity holder in publicly-traded company, Honoraria. Yoshino:Kyowa Kirin: Honoraria; Chugai Pharmaceutical Co., Ltd.: Honoraria, Research Funding. Maeda:AstraZeneca: Research Funding; Astellas: Honoraria, Research Funding; Chugai Pharmaceutical Co., Ltd.: Honoraria, Research Funding; Nippon Shinyaku Pharmaceutical Co., Ltd.: Honoraria, Research Funding; Novartis Pharma: Research Funding, Speakers Bureau; Otsuka Pharmaceutical: Honoraria; Eisai Pharmaceutical Co., Ltd.: Honoraria; Kyowa Kirin: Honoraria; Takeda Pharmaceutical Co., Ltd: Honoraria. Ennishi:Kyowa Hakko Kirin Co., Ltd.: Honoraria; BMS: Honoraria; Ono Pharmaceutical: Honoraria; Janssen: Honoraria; Incyte: Honoraria; Eisai: Honoraria, Research Funding; Chugai: Honoraria, Research Funding; Nippon Shinyaku Co.: Honoraria, Research Funding.